An Ode to Basic Research
How we got vaccines in record time
When COVID first hit, the realistic "best case" timelines for treatments or vaccines were at least 12-18 months. The biopharma industry typically takes 5-7 years and billions of dollars to produce a new medicine. More ambitious timelines of 9-12 months were proposed, but an analyst described this scenario akin to "throwing a dart 40 feet away blindfolded and hitting a bullseye."
What was unthinkable last March has succeeded: tens of millions of people have been vaccinated using a new technology (mRNA), and several other vaccines using different technologies are about to launch.
The speed of this achievement has had a polarized response. Was this a rushed and unsafe process? Dumb luck? Or are cures sitting around waiting to be made? Given that COVID affects everyone, and we went from no treatments or vaccines to multiple in less than a year, it’s worth diving into how the industry pulled it off.
What if I told you we weren’t blindfolded at all?
Though new platforms like mRNA can speed up the vaccine design process, they’re useless without a target to shoot at (i.e an antigen). Prior to the emergence of COVID-19, scientists had already determined a highly effective antigen for coronaviruses. Without a viable antigen, attempts to speedily create a vaccine would have failed.
What we're seeing today is made possible by:
Decades of research
Individuals risking their careers to chasing discoveries that may never work
Public/private sector willingness to fund projects with a very high failure rate
... all in pursuit of the huge breakthroughs when something does work.
In this post I’ll use the story of COVID vaccines to explain the role “basic research” plays in decoding the natural world and in the creation of new medicines.
Where do medicines come from?
Framework and illustrations No Patient Left Behind:
Before getting into the science, I’ll first introduce our cast of characters. There is a virtuous cycle between academia, startups, and large biopharma, funded and supported by the government and private foundations. This post focuses on the foundation of basic research, but each player had an important role in this story.
Basic research — the foundation
Virtually all the technologies that power modern life are built atop a foundation of basic research. Basic life science research seeks to fundamentally understand the mechanics of natural phenomena, and to determine how these phenomena can be manipulated or used. The focus is more about increasing knowledge than creating products.
A basic scientist achieves that goal by publishing papers in academic journals that increase the world’s knowledge in a particular domain (beautifully illustrated here). Their career, prestige, and reputation rests on the novelty of their discovery and how widely cited their work is. When you think of basic scientists, picture PhD students, post docs, and professors at universities working in labs and writing grants. Nobel Prizes in medicine are typically awarded to basic researchers, where they discover something fundamentally important to how the natural world works, but the commercial implications of their discovery are not realized for some time.
Trying to discover new things about the world is lengthy and often futile. A scientist can spend 5-10 years of their life on a discovery that does not pan out, or is ultimately trivial. Kaitlin Kariko is one such scientist, who got demoted during her relentless pursuit to prove mRNA could be a viable therapeutic. Her work was deemed insignificant by her superiors. In hindsight, it seems obvious that her work was important, but in basic research it’s both difficult to know if something is viable or valuable. If the commercial viability or relevance of discoveries is difficult to know, where does the money come from?
Basic research is funded by groups who do not, strictly speaking, need a return on investment. This largely means the federal government (led by the National Institutes of Health) and is supplemented by private foundations (i.e. HHMI). Basic research does not mean completely detached from practical utility: there are often focus areas and priorities. For example, the National Institute of Allergy and Infectious Diseases led by Anthony Fauci, or the National Cancer Institute. Each funds and conducts research on the underlying mechanisms of their respective disease areas, in the hopes that this knowledge will eventually lead to new treatments and cures.
Startups — the risk/reward of commercializing discoveries
In order to translate discoveries made in a lab to treatments given to patients, scientists often form venture backed startups to commercialize basic research.
Moderna and BioNTech were startups formed around the idea that mRNA could be used as a therapeutic in humans, based on the discoveries made by Kaitlin Kariko.
It’s not always clear what that killer app of a discovery will be. Much of the original focus on mRNA was to treat cancer, rather than infectious diseases. New modalities or platforms can fail to work for decades, be declared dead, only to ultimately succeed.
Big BioPharma — bringing discoveries to market
Large biopharma are constantly on the hunt for the next big thing. There is constant pressure to either internally develop new medicines, or partner with or buy other companies to acquire new technologies.
The process of bringing medicine from “bench to bedside” is complex, and requires expertise in development, manufacturing, clinical trials, and distribution. A startup or academic institution usually does not have all these capabilities built out, and thus need to partner with a larger company to get medicines to market. Pertinent examples include BioNTech/Pfizer and Oxford/AstraZeneca. In exchange for being able to profit off of the medicines they develop and distribute, their patents expire and their medicines go generic, yielding a huge societal ROI.
Big biopharma and startups are the most visible and widely known players in this ecosystem. However, fundamental discoveries in basic research allow them to stand on the shoulders of giants. The better we understand the natural world, the greater our ability to use its phenomena to benefit humanity.
The wonder of adaptive immunity
Right now we’re fixated on one pathogen: COVID-19. But our world is filled with bacteria and viruses. A teaspoon of soil contains on the order of a billion bacteria! While not all of these are actively harmful, for organisms to survive they need to be able to defend themselves from pathogens.
The marvel of our immune system is that not only can it defend us from pathogens it has never seen before, it can also develop a memory to prevent re-infection in the future. Scientists were able to reach this conclusion hundreds of years ago without knowing a lot of the underlying mechanics of how this worked. As far back as 1778, scientists determined that exposing people to (less harmful) cowpox could vaccinate against smallpox.
When I was a freshman in college, I had an “aha” moment where I realized even today how much we know about symptoms and treatments, but how little we know about everything in between. I was meeting with a professor who was studying what actually happened after anthrax infected you. I was confused, because I figured we must know how that works if we already had an antibiotic to treat it. The goal of his work was to understand the underlying mechanics of infection so that better treatments could be devised. Basic research reveals and details the underlying mechanisms of biology, leading to new theories and opportunities for therapeutics.
Vaccines = effective antigens for a pathogen
As our knowledge of the immune system has expanded, we have been able to take a more precise, engineered approach to creating vaccines. A key component of this is understanding for a given pathogen, which antigen will lead to a successful vaccine. An antigen is simply a part of a pathogen that produces an immune response.
You know when you login to a website and you need to prove you’re not a robot? They’ve gone from “find the motorcycle” to “find all the squares that have a motorcycle in it.” These CAPTCHAs aren’t just to prevent malicious logins, they’re meant to train AI to be able to precisely recognize individual features in the images. This is analogous to how your immune system works.
In this image, imagine the motorcycle as the invading “pathogen.” As it infects your body or is attacked by the immune system, it’s broken up into different recognizable pieces, like the different squares. Developing vaccines is the art and science showing the right antigens of a pathogen to prompt your immune system into generating an enduring defense. The antigen that successfully leads to effective immune response is a mystery that must be solved for each kind pathogen — these things evolved to infect you!
An antigen you’re likely most familiar with is a weakened or dead virus. That’s akin to showing your immune system the whole motorcycle. While that works for several kinds of pathogens, there are many others where it’s not an effective antigen. Most new vaccine platforms are some form of showing the right subunit or piece of the pathogen, to generate protection from infection in the future.
Basic research already identified effective antigens for coronaviruses
Six years of work on SARS and MERS prior to COVID-19, led by Kizzmekia Corbett
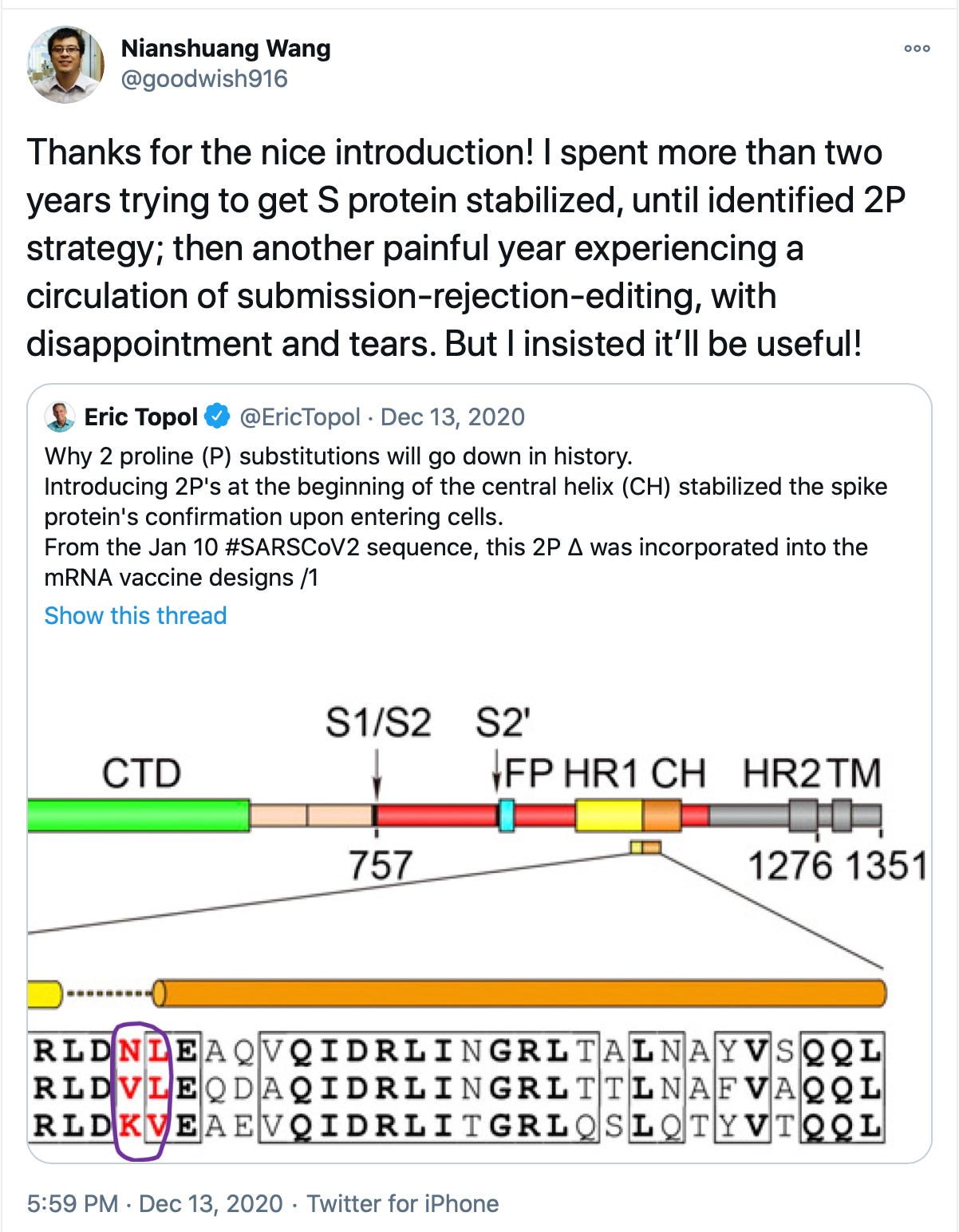
One of the biggest reasons vaccines were created so quickly was that a viable antigen had already been identified. We had prior pandemic scares with SARS and MERS that essentially disappeared. Fortunately for us, years of publicly funded basic research explored how these viruses worked, and determined what antigens to target should they ever return. This was pandemic preparedness that actually worked. Scientists identified the “spike protein” as a viable antigen, and also figured out exactly how to produce a stabilized form of it.
The importance of this type of discovery is not always appreciated at the time! The paper was rejected 5 times before being published in 2017. Without this work, attempts to speedily create a vaccine would have failed.
Basic research produced new vaccine platforms
Same antigen, different vaccine platforms
Much of the buzz around vaccines has focused on the technologies powering next generation vaccine platforms. These platforms are unique in that instead of manufacturing an antigen in a factory, the vaccine is providing instructions to your cells to produce the antigen.
Creating the instructions to produce an antigen can be done faster than manufacturing the antigen itself. Combine these new platforms with known antigens and you’ve got a recipe for speed.
Being humble in this triumph
"Evolution is cleverer than you are.” -- Orgel’s second rule
In many ways we’re lucky COVID-19 was the pandemic we got. That statement might sound crazy given the carnage it has wreaked.
Scientists already had a strong candidate antigen for an effective vaccine, and had developed next generation vaccine platforms that could design vaccines quickly. In addition, research indicates coronaviruses do not dramatically mutate to evade the immune system the way that other viruses do. This hopefully means the vaccines are likely to be effective for some time, rather than needing a new one each year.
In contrast, after $76b and decades of research, there is still no vaccine for HIV. This is not for lack of effort, priority, or funding — this pathogen is a particularly cunning enemy. The investments into understanding and decoding HIV helped dramatically increase the knowledge of how many other viruses work, and led to the creation of antivirals and vaccine technologies. Failed attempts at creating an HIV vaccine paved the way for success for an Ebola vaccine, and now COVID vaccines.
The COVID vaccine race brought together every player in the biopharma ecosystem: government agencies, academia, startups, and large biopharma companies. Basic research provided a viable antigen and vaccine platforms built for speed. With this foundation in place and a global emergency, the biopharma industry developed not just one, but many vaccines.
Thank you Shaan, Agnes, and the community at Compound Writing for the feedback and edits.The inspiration for this post came from a course I took and these tweets.
Footnotes for anyone still reading
CRISPR is actually just the immune system of bacteria. We have engineered it to be a gene editing tool in humans.
You might be wondering why not make the vaccine the old fashioned way? Safety, speed, efficacy
The adenovirus vaccine platform failed to work in HIV, and in fact might have made people even more susceptible to the disease. It was successfully used to create an Ebola vaccine. Both Oxford/AstraZeneca and Johnson & Johnson are using this technology for their COVID vaccines and seem likely to succeed
High tech success followed by low tech bumbling. I’d love to learn more about why the U.S. is great at certain things and terrible at others
Variants: Vaccinating people isn’t just about saving an individual from infection, it’s reducing the chances of new variants. The more people who get infected and the longer this goes on, the greater the likelihood of new variants emerging. Variants have already emerged that are more infectious with the same lethality, netting a significantly higher death toll. Vaccines are likely to be effective against these variants, but they could evade the treatments that have been developed.










One never knows what basic research will pay off. I remember the 1982 Nobel Prize was for discovering prostaglandins, but that was only possible because in the 19th century the Prussian government was paying some scientist to look up mole assholes and measure their prostate glands to look for seasonable variance. I'm not going to ask what prior research led to that project. I don't want to know.
Great overview of the bench-to-bedside process leading up to current vaccines. I'd argue, however, that even if we didn't have SARS, MERS, etc results to inform SARS-CoV2 research, the mRNA platform would have still drastically cut down on time to first dose. Once you know the viral genome, you can model the antigen (albeit may not perfectly), and then develop the vaccine.
Also, if you're interested in learning more about mutations (i.e. antigenic shift and antigenic drift), here's a shameless plug for my substack:
https://amrchairmed.substack.com/p/sars-cov2-mutations?r=4raep&utm_campaign=post&utm_medium=web&utm_source=copy